15: Green Revolution
- Page ID
- 8286
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)In the last few chapters, we have seen Americans spread across the continent. Beginning with the original thirteen colonies, the history of the United States often seems to be a straightforward story of expansion westward. After a Revolution motivated partly by the colonists’ resistance to the Crown’s restrictive Proclamation, citizens of the new nation took full advantage of opportunities provided by the Northwest Ordinance, the Louisiana Purchase, the Mexican-American War, and the Oregon Treaty, to seek their fortunes on the frontier. Expansion was achieved partly by hardworking farmers, ranchers, and miners on the countryside, and partly by urban wage-workers and businesses in new western cities. These pioneers were aided by capital from eastern and overseas financial markets, by improving transportation networks, and by eastern city workers and capitalists who processed and consumed the products of fields, forests, and mines.

But not all the significant movement in American History was east to west. The growth of eastern cities depended on resources acquired on the frontier, so the flow of raw materials like farm products, timber, and minerals (which we will examine in Chapter Twelve) from west to east was vitally important to the nation’s growth. The ideal of Manifest Destiny was as much about securing access to these resources as it was about planting the stars and stripes from sea to shining sea. And sometimes, important changes actually originated in the west, and expanded eastward.
The expansion and growth of both the West and the East depended on the ability of farmers to continue producing food for the nation’s growing population and for export. The biggest single factor in the continuing farm bonanza was soil fertility. In the 1840s, German chemist Justus von Liebig discovered that the chemicals nitrogen, phosphorus, and potassium were essential to plant growth. Farm soils naturally lose fertility unless fields are given time to regenerate between crop plantings or amendments are added. Liebig announced that instead of leaving fields fallow for years between crop plantings, farmers could amend the soil with the chemicals plants needed. We will examine each of these chemicals, beginning with the element often considered the most important soil amendment, nitrogen. During the crucial period when America’s commercial farmers became increasingly responsible for feeding the growing nation, nitrogen came from the west.
Green Manure, Guano, Caliche
When early settlers cut and burned forests to build farms, the thin soils of the forest floors received an initial productivity boost from leaf mulch and from the ashes of burned trees. This fertility was exhausted in a few years unless the farmer rotated crops and pasture or added manure to the soil. The prairie soils later settlers found farther west were deeper and more fertile than eastern forest soils. Tallgrass prairies grew from the Canadian border to Texas, and as the name suggests, some of their grasses grew six to nine feet tall. When Laura Ingalls’s parents told her not to stray too far onto the prairie in the Little House books, it was because they were afraid of losing her in the tall grass.
In addition to being tall, the prairies were deep. After millennia of growth and decay, prairie vegetation had left a layer of humus up to ten feet deep. The roots of perennial grasses took advantage of this depth, drawing water and nutrients from well below the surface. When settlers arrived with their new John Deere cast-steel plows, they only managed to turn over the top foot or so of this organic layer. But even so, the prairie offered some of the richest soils American settlers had ever seen. Western farmers wrote their families and friends back east boasting of the land and the great yields they got farming it.
In the long run, even the most fertile soils will be depleted after years of growing annual crops that don’t return any nutrients. Smaller farms were able to spread manure from livestock, but as their cultivated fields grew from a few acres to a few hundred, most farmers found there was just not enough manure to spread. Commercial farmers, who specialized in growing wheat or corn for the market, began planting their fields with the cash crops every year rather than rotating grain planting with pasture planting for grazing animals that would return at least some manure to the land. A new source of fertility was needed to prevent the agricultural balloon from deflating. The solution was alfalfa, a crop farmers called Chili Clover.

It has long been known that in addition to spreading manure, planting legumes is an effective method of returning nitrogen to the soil. Although nitrogen is the most common element in Earth’s atmosphere, atmospheric nitrogen is very stable and unreactive. Plants cannot use nitrogen until it has been converted to nitrogen oxides, nitrates, or ammonia. Legumes have evolved a symbiotic relationship with Rhizobium bacteria which live in nodules on the plants’ roots and break the bonds of atmospheric nitrogen. When legumes are grown on a field and then plowed under, the nitrogen accumulated in the plants enters the soil and becomes available for the next crop. One of the most effective legume crops used as a green manure is alfalfa.
Alfalfa is native to Eurasia, where Siberian horse tribes began feeding it to their animals over three thousand years ago. Cortés and Pizarro brought alfalfa to Mexico and Peru in the early 1500s and the aggressive species quickly spread to across Central and South America. When English colonists planted alfalfa, which they called lucerne, the plant did poorly in the cool, acidic soils of the Atlantic colonies. After this early failure the plant was forgotten by American farmers. But a Chilean miner introduced alfalfa to California during the Gold Rush. The climates of Chile and California are nearly identical, and the Chili Clover thrived in western pastures, renewing soils and providing ten times the nutrition of regular grasses for the growing dairy herds of California. Impressed by alfalfa’s success in California in the early 1850s, the U.S. Patent Office mailed out thousands of packets of alfalfa seed to farmers across the country. Alfalfa plantings spread eastward from California as the plant was quickly adopted by farmers in Utah, Colorado, Kansas, and Nebraska. Today, the Chilean import is so common in American fields, almost no one remembers it was not a native species.
But some farmers lacked the patience to rotate cropping with pasture, or were unwilling to accept the idea that a significant portion of their farms would be fallow each year. They preferred to amend their soils rather than waiting for fertility to regenerate naturally. But as mentioned, few farmers had access to enough manure to supplement all their soil. The first commercial fertilizers were made from guano, the droppings of seabirds living on islands off the western shores of South America. Guano comes from the Quechua Indian word Wanu, which means any excrement used as a soil additive in farming. The advantage guano had over locally-available manure was that it was dry, light, and highly concentrated. Natives of the Andes have mined guano on the coast and islands for at least 1,500 years, according to archeologists. Spanish colonial records noted that Inca rulers considered protecting the cormorants that were the main source of guano so important, that disturbing the birds’ nesting areas a capital offense. Guano was carried from the coast up into the Andes on the backs of llamas, for use on the terraced farms surrounding highland cities like Machu Picchu.

Although surrounded by ocean, the islands off the western coast of South America are arid. Like the deserts they face on the mainland, some experience no annual rainfall at all. Seabirds such as cormorants and pelicans have lived on these islands by the millions, for thousands of years. Over that time, they’ve left literal mountains of droppings, which due to the lack of rain have simply piled up. The guano contains 8 to 16 percent nitrogen, 8 to 12 percent phosphorus, and 2 to 3 percent potassium, which makes it an excellent fertilizer without any mixing. It simply needs to be chopped off the mountain, ground up, and spread on fields.
Prussian explorer Alexander von Humboldt visited the islands around 1802, and publicized guano’s value as a fertilizer throughout Europe. Seeing a lucrative business opportunity, Europeans and Americans fell on the area in a guano rush, and by the middle of the century several nations had enlisted the work of Chinese peasants in a Pacific labor system that has been compared with the slavery of the Atlantic world. Although the Chinese workers were technically free, many had been tricked into labor contracts promising work in California. Once they reached the guano islands and realized they had been duped, there was no way off. Over a hundred thousand Chinese workers were imported to the islands in the second half of the nineteenth century.
Guano was so profitable that the U.S. Congress passed a Guano Islands Act in 1856. The law provided an incentive for American sailors to find and claim undefended islands for America by giving the discoverer exclusive rights to the guano recovered. Islands claimed under the Guano Islands Act include parts of the Hawaiian chain, Midway Atoll, part of American Samoa, and several islands still disputed with Colombia. The guano islands off the western coast of South America were so valuable that two wars were fought over them. Chile and Peru fought Spain in the Chincha Islands War, 1864-66, and defeated the Spanish Empire. Then, Spain’s claim had been successfully set aside, Chile took many of the guano islands from Peru, along with the nitrate fields of the Atacama Desert, in the War of the Pacific, 1879-83.

After about 1870, guano was overtaken as a source of nitrogen by nitrate-rich desert soils called Caliche. These soils were discovered in the Atacama desert, a region regarded as the driest place on earth that lay partly in Chile, partly in Peru, and partly in Bolivia. When all three nations rushed to extract and process the Caliche, Chile challenged its northern rivals for the nitrate fields. Chile’s victory in the War of the Pacific extended its border northwards to encompass the Atacama desert, including all the coastal territory that had belonged to Bolivia. Many ethnic Bolivians living around the port city of Arica still dream of throwing off the Chilean yoke and winning their country access to the Pacific again.
Defeating its northern neighbors in the War of the Pacific made Chile the undisputed power on the west coast of the Americas and generated an economic boom. The nitrate Chile monopolized was valuable both as a fertilizer and as a key ingredient in explosives and munitions. But mining and processing nitrate from Chile’s desert soil required much more capital than digging guano. Chile attracted British investors, and soon joint ventures began shipping a million tons of nitrate per year out of the South American desert. Production grew steadily until 1914, when World War I created new incentives for Britain’s enemies to find an alternative to Caliche nitrate.
Fritz Haber was the German chemist who in 1911 developed the high-pressure method of extracting nitrogen from the atmosphere that is used today to produce nearly all the nitrogen used in industry and agriculture. Carl Bosch, working for German chemical company BASF, scaled up Haber’s laboratory experiment to industrial production. In 1914, BASF produced 20 tons a day for the war effort. After the war, production of synthetic nitrate fertilizer quickly cut prices by more than half, and annual Chilean caliche shipments dropped to less than a third of their 2.5 million ton pre-war volume. Haber and Bosch were each awarded Nobel Prizes for their work in chemistry.
The Haber-Bosch process requires not only extreme high pressures to extract atmospheric nitrogen, but a great deal of energy. Approximately five percent of the world’s production of natural gas is required to produce about 500 million tons of ammonia each year. But the results have been spectacular. The application of concentrated nitrogen to farm fields increased production even over the yields that had been achieved using guano and nitrate, causing a global explosion of crop yields known as the Green Revolution. By 2010, the Haber-Bosch process produced 133 million tons of ammonia annually, about three quarters of which was applied as fertilizer. Corn yields per acre increased five-fold, and other staple crops such as rice saw increases of three or four times. Historian of technology Vaclav Smil estimates that if crop yields had stayed where they were in 1900, by the year 2000 farm fields would have needed to cover at least half the land on the ice-free continents in order to feed the world’s population, rather than the fifteen percent they now occupy. Another way of putting Smil’s point is that nearly half the people alive today would probably starve, without the Haber-Bosch process.

Although he won a Nobel Prize and worldwide admiration for his work leading to nitrogen fertilizers, Fritz Haber’s legacy is clouded with tragedy. In addition to the nitrate Germany required for munitions, Fritz Haber’s other contributions to the German war effort were the chlorine-based chemical warfare agents used against allied troops in World War I and a cyanide-based pesticide called Zyklon A that was a direct predecessor of the poison used against prisoners in the concentration camps during World War II. Haber’s wife, Clara Immerwahr, was also a scientist and was the first woman ever to receive a Ph.D. from the University of Breslau. Immerwahr killed herself with her husband’s gun when she discovered he had supervised the first successful use of chlorine gas at the battle of Ypres in 1915. Their son Hermann emigrated to the U.S. and committed suicide in 1946 after discovering that his father’s invention had been used to kill millions of their fellow German Jews. Fritz Haber did not live to see Zyklon B used in the concentration camps. He died while moving to Palestine in 1934.

The Haber-Bosch process is an effective way to synthesize nitrogen as long as energy costs remain low, but it is not the only way to make nitrogen fertilizer. As mentioned earlier, nitrogen fixating soil bacteria and symbiotic microbes living on legumes fix nitrogen from the atmosphere without any additional energy inputs. And there are other chemical processes that can be applied on industrial scales. Some of these industrial processes are much less energy-intensive than the Haber-Bosch. For example, a hydro-electric plant in Norway used its surplus energy to make ammonia for fertilizer from 1911 to 1971, using a process that had actually been developed before Fritz Haber made his discovery. But in spite of these alternatives, almost all the nitrogen used in commercial fertilizer is currently produced through the Haber-Bosch process. As a result, fertilizer prices tend to follow natural gas prices, and ammonia-based fertilizers will probably continue to be made using the high-energy process until energy costs rise substantially.
Phosphorus and Potassium
The second element Liebig discovered was crucial to plant growth is phosphorus. Unlike nitrogen, which makes up seventy-eight percent of Earth’s atmosphere, phosphorus is a mineral. Traditional agriculture recycled phosphorus by returning manure to fields as fertilizer. During the green revolution, most of the phosphorus applied by the world’s farmers has been superphosphate, a concentrate manufactured from a raw material called phosphate rock. Phosphate rock is a sedimentary mixture of minerals deposited in earlier geological eras on the bottoms of ancient oceans. Guano was also rich in concentrated phosphate, and after the Atacama Desert became the main source of nitrates, much of the guano remaining on the Pacific islands was used to produce phosphate fertilizer. The deposits currently used to make fertilizer contain about thirty percent phosphorus.
Although mineral phosphorus is plentiful in the Earth’s crust, known deposits of sedimentary phosphate rock are finite. Estimates of the concentrated phosphorus remaining vary, and depend on assumptions about yet to be developed technologies for discovering and recovering the mineral. Some scientists believe phosphorus production may peak around 2030, and then decline as exploitable reserves are exhausted. Like nitrogen, inexpensive and plentiful phosphorus has boosted crop yields greatly. Eighty percent of the phosphate rock mined each year goes to produce fertilizer. Phosphorus shortages would create serious threats to world food supply, which could impact global security. But since phosphorus is still available in animal and human waste, a shift back toward applying manure to agricultural fields would greatly reduce demand for superphosphate. Recognizing the growing value of the chemicals contained in our waste, some urban sewage treatment operations have begun producing concentrated phosphorus fertilizers from city sewer sludge.

Potassium, the third vital plant nutrient, is actually named after the potash that early American colonists made from trees they burned to clear their fields. Ashes were soaked in water, and then the water was evaporated in cast iron pots to produce lye (potassium hydroxide) and potash (a mixture of potassium chloride and potassium carbonate). Wood ashes yielded about ten percent potash by weight, and by clearing a few acres of trees each year as they expanded their cultivation settlers could also produce a product they could sell for cash. As previously mentioned, the first patent issued by the United States in 1790 was for a “new apparatus and process” for making potash.
Like phosphorus, commercial potassium fertilizers are manufactured from sedimentary deposits mined at the sites of ancient inland seas. Current world production is dominated by two cartels, but known reserves total more than three hundred times annual production, so the element is not considered strategic.
Hazards
One of the problems associated with using concentrated soluble nitrogen, phosphorus, and potassium (NPK) in agriculture is that uptake by crops is less than 50 percent. In the old days of ten-foot deep prairie soils, this would not have been a problem. Organic matter binds elemental nutrients, and then decays slowly and releases the nutrient chemicals into the soil gradually over many seasons. The deep root systems of perennial grasses stored nutrients in biomass. And the chemistry of organic soils and the microscopic insect, bacterial, and fungal life-forms that thrived in those soils held onto the rest. However, in the shallow-plowed fields of modern farms, the root systems of annual crops are not deep enough to catch and hold nitrogen, phosphorus, and potassium as it runs off with the water. And much of the soil life that once thrived in farm fields and pastures has been eliminated by pesticides, herbicides, and fungicides.
In spite of the additional food produced to support growing world population, the green revolution has been criticized by environmentalists for ignoring the problems created by fertilizer runoff. Excess agricultural fertilizer contributes nearly half of the nutrients that are currently poisoning American streams and rivers. Fertilizers entering the watershed encourage algal blooms. When these prolific mats of single-celled plants die, the decaying algae traps dissolved oxygen, creating hypoxic dead zones. The dead zone in the Gulf of Mexico has shrunk somewhat from its 2002 peak of 8,500 square miles. But excess fertilizer continues to flow down the Mississippi into the ocean.
Video \(\PageIndex{1}\): A YouTube element has been excluded from this version of the text. You can view it online here: https://youtu.be/rK_mEHqx7rw. Video from NOAA, Public Domain
The green revolution has also been criticized by less developed nations for increasing poor people’s dependence on technological solutions created in the faraway laboratories and factories in the United States and other industrialized nations. In many cases, development loans from organizations like the World Bank are tied to spending on products from the donor nations. Critics argue this benefits the donors more than the recipients and puts poor farmers at risk when they become dependent on new technologies and then fail to earn enough with their crops to pay rising prices for seeds, fertilizers, and pesticides. Farmers in developing nations have been encouraged with generous loans to invest in expensive capital equipment they cannot afford when the loans disappear. Since the 1990s, more than a quarter million farmers in India have killed themselves because they cannot escape the cycle of debt created by their green-revolution involvement in high-tech agriculture.
Dust Bowl
In America, the early green revolution that began with the use of guano, potash, and nitrate led to bonanza farming and the extension of agriculture onto marginal lands. The Russian Revolution and World War I at the beginning of the twentieth century reduced European farm production and drove up the price of American grain in international markets. Marginal high plains grazing lands like those of western Kansas and Nebraska, Oklahoma and northwestern Texas were put under the plow. Cropland in this region doubled between 1900 and 1920, and then tripled again between 1925 and 1930.

Plowing fields for annual corn and wheat planting is such a common farming practice, it seems normal. On the high plains, perennial grasses had evolved to find water deep in the soil and hold onto it. Plowing exposed the soil to the sun and wind, and cut the roots that trapped moisture and bound the soil together. Worse, generations of American farmers who had been taught to depend on plentiful commercial fertilizers, had forgotten the value of humus. Since Justus von Liebig had focused their attention on soil chemistry in the 1840s, most soil scientists had promoted the idea that chemical nutrients were the important thing, and that soil itself was just an inert medium. Although the prairies settlers discovered when they moved westward had been rich in living organic material, the farms built on those original soils relied on imported chemicals rather than on nutrients produced by biological activity in the soil. Many farmers even burned off the stems and stalks left after each harvest. Then the soil blew away.
The western edge of the prairie was actually a completely different ecosystem from the eastern edge by the Great Lakes, but the change in climate was very gradual. Although many farmers moving west to convert the high plains rangeland to cultivation failed to notice it, rainfall was scarcer and the wind blew harder. Those who did notice were mollified by the assurances of experts that “rain followed the plow.” But of course that was just boosterism and wishful thinking: rain did not follow the plow. Between 1933 and 1935, drought struck the area. Actually, dry conditions returned after a few years of unusual wetness that had been taken by hopeful farmers and optimistic boosters as the region’s permanent climate. Over half a million people were left homeless when their topsoil blew away.
In a single storm, beginning on November 11, 1933, topsoil from Oklahoma was blown all the way to Chicago, where over 12 million pounds of it fell on the city like snow. Like alfalfa, guano, and nitrate in the nineteenth century, America’s topsoil was traveled from west to east. On Black Sunday, April 14, 1935, dust storms were reported from the Canadian border to Texas. Newspaper reporters throughout the affected area wrote that they could not see five feet through the blowing dust. The agricultural disaster that became known as the Dust Bowl caused an exodus from the high plains region that should never have been put under the plow. But the disaster was not just just agricultural. Of 116,000 refugee families surveyed on their way into California, only four out of ten were farmer families. A full third of the heads of families who fled Oklahoma, Kansas, Nebraska, and Texas were white collar professionals. When the farms blew away the whole region was wiped out.

Alarmingly, less than a century later the lessons of history have been forgotten and much of the Dust Bowl region is again under the plow. The wheat-fields of Kansas, Texas, Oklahoma, and Colorado produced 411 million bushels of grain in 1933, just before the drought began. The Dust Bowl disaster cut production by three quarters, but by 2012, the region was once again producing 700 million bushels of wheat. This time, farmers have taken advantage of irrigation.
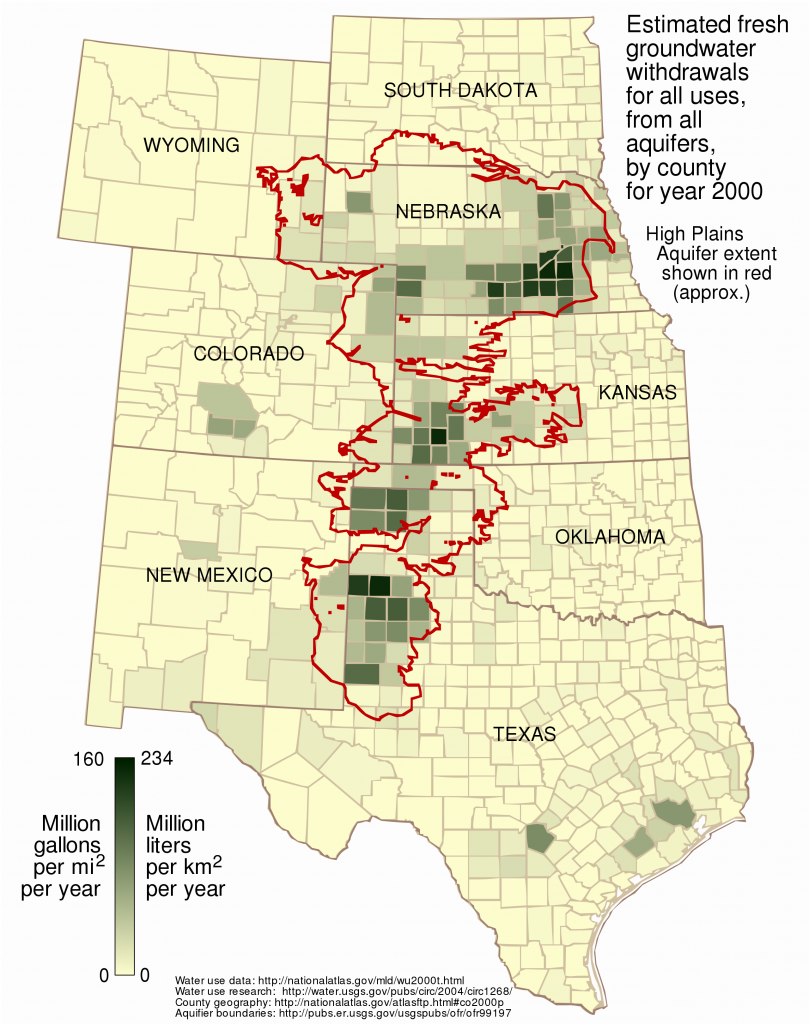
The Ogallala is one of the world’s largest aquifers, stretching from South Dakota to West Texas. Farmers have been drawing water from it for over fifty years. But the aquifer, which was charged with water over centuries before American farmers arrived, is not an unlimited resource. Sections of West Texas and Kansas have already run out of water, and irrigation is still accelerating. In just seven years between 2001 and 2008, farmers used about a third of the water that was taken from the aquifer during the entire twentieth century. The rate of depletion far exceeds the rate of recharge, even in the occasional rainy years. If the aquifer is emptied and irrigation becomes impossible on the high plains, American grain production will be reduced by 700 million bushels per year. Farmers will have no choice but to replant perennial prairie grasses and convert the plains back to rangeland, if they hope to prevent the soil blowing away again. The question is, will American agribusiness change willingly and thoughtfully before a crisis, or out of desperate reaction after the crisis strikes?
Further Reading
- Edward Dallam Melillo, Strangers on Familiar Soil: Rediscovering the Chile-California Connection. 2015.
- Vaclav Smil, Enriching the Earth: Fritz Haber, Carl Bosch, and the Transformation of World Food Production. 2004.
- Donald Worster, Dust Bowl: The Southern Plains in the 1930s. 1979.


